READYCELL KITS APPLICATIONS
Blood Brain Barrier Assays
The use of our products will let you assess the permeability and the transporter-drug interaction of this challenging barrier, the BBB.
Worldwide shipments at room temperature thanks to our patented technology.
Full cell functionality after transportation.
No in-house cell culture maintenance nor cell licensing costs.
BBB MODELS
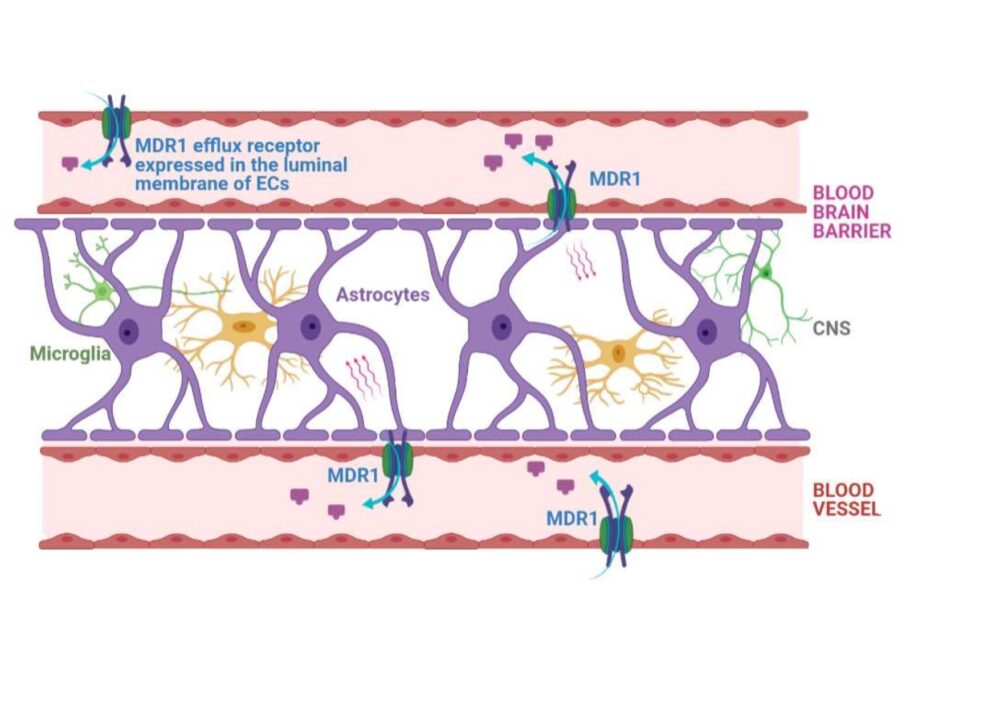
Ready-to-use efflux proteins for BBB assays
MDR1 expression
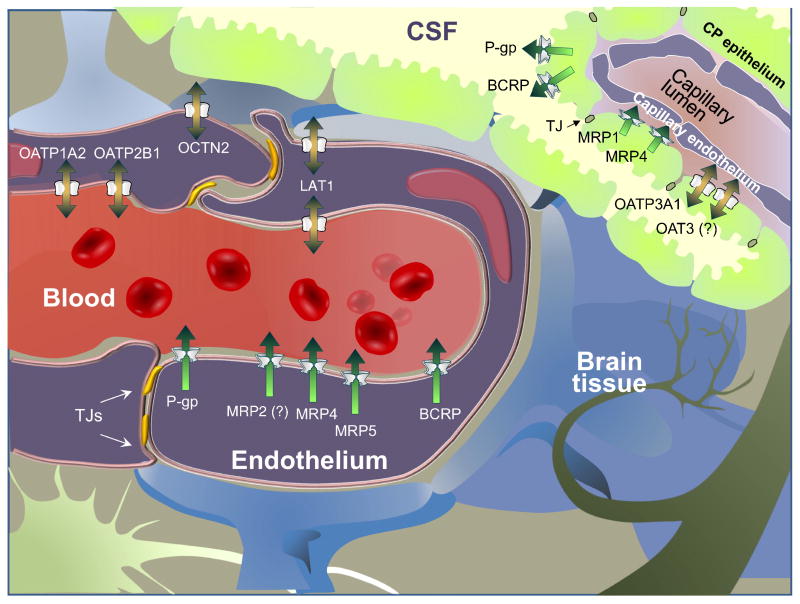
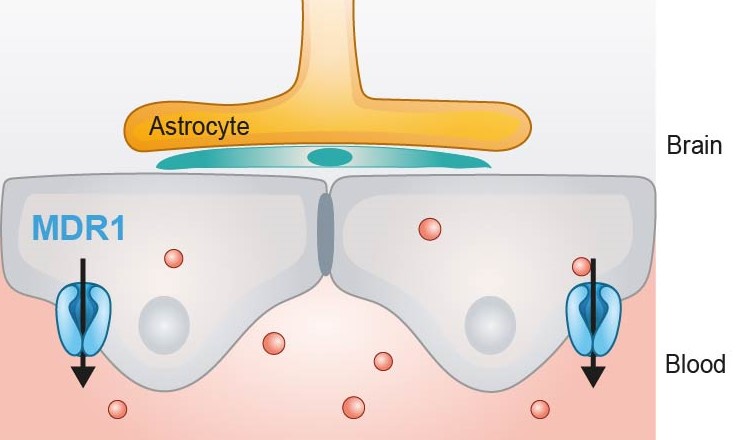
The multidrug resistance protein 1 (MDR1) also known as P-glycoprotein (P-gp) is a a membrane efflux protein which needs from the ATP hydrolysis to actively translocate substrates against an electrochemical gradient. They are present in our CacoReady and PreadyPort MDR1 Kits.
Read more
This efflux transporter is expressed in tissue barriers (e.g brain, intestine, placenta) and excretory organs (kidney and liver) where it exerts a protective mechanism by pumping out of the cells and into the blood, bile and urine compounds potentially toxic.
In particular, in the brain, MDR1 is expressed in the apical membrane of endothelial cells where it avoids drug accumulation in the brain enhanced by its broad substrate specificity. Blocking of this efflux transporter alters drug pharmacokinetic parameters and tissue distribution of drugs, increasing drug half-lives and toxicity effects. In fact, the most pronounced pharmacological effects of MDR1 inactivation were seen in the brain, which indicates the important role of MDR1 to prevent compound permeability in the CNS.
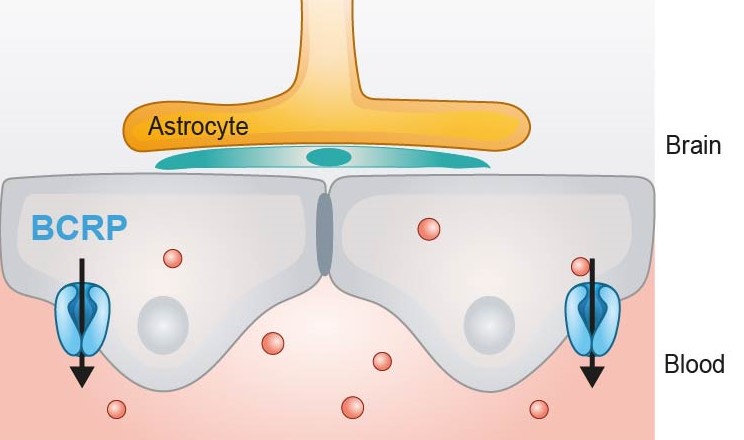
BCRP expression
BCRP efflux transporters are also located at the luminal membrane of the BBB and recognizes a broad number of endogenous and xenobiotic compounds, that overlap with those of Pgp. They are included in our CacoReady and PreadyPort BCRP model.
Read more
Like MDR1, the breast cancer resistant protein (BCRP) is an efflux transporter that belongs to the ATP-binding Cassette Family (ABC) family. Despite BCRP is expressed in a wide number of tissues, there is evidence that this transporter is relevant as one of the major drug transporters involved in clinically relevant drug disposition in the CNS.
BCRP which is more prominent express in humans than MDR1 is found to work cooperatively with MDR1 to restrict compound penetration into the BBB. Therefore, it may happen that the inhibition of BCRP does not prevent the permeability through the BBB of those molecules that are co-substrates of both transporters.
ReadyCell Blood Brain Barrier kits
We help you determine the suitability of your compound to cross the BBB
It is recommended to assess the in-development molecule interaction to P-gp and BCRP receptors to understand the pharmacological route and optimize clinical stages.
CacoReady, and PreadyPort MDR1 and PreadyPort BCRP are three assay-based kits that can be used to identify whether new molecular entities are MDR1 or BCRP substrates/inhibitors.
Blood Brain Barrier ANATOMY
Characteristics of the blood brain transmembrane
READYCELL Support
Frequently Asked Questions
How does the permeability of the BBB differ from that of intestinal barrier?
Blood brain barrier is composed by different cell types (endothelial cells, perycytes and astrocytes). Endothelial cells which line up the brain capillary blood vessels are polarized and present efflux transporters and poorly characterized uptake transporters at the apical and basal membrane which aids BBB to maintain brain homeostasis. It has been reported that, unlike intestinal barrier, BBB prevents the diffusion of > 98% of small-molecule drugs and most of the large biologic therapeutics.
Is CacoReady a representative model of the BBB?
CacoReady model is a good alternative to study BBB permeability despite their epithelial origin since Caco-2 cells form a tight barrier with an electrical resistance above 1000 Ω x cm2. In addition, polarized Caco-2 cells also express the MDR1 and BCRP efflux transporters that are present in the luminal site of the brain endothelial cells.
Is the quality control data specific for each kit?
Yes, we provide a concrete quality control for each batch so the customer can have a reference for the kit both in pre-shipment and post-shipment.
Does ReadyCell shipping medium affect cell lines?
No, ReadyCell’s Shipping Medium consists of a semi-solid culture system specifically designed to preserve cells at room temperature (15-25ºC). This medium maintains a suitable physicochemical environment, keeping adequate moisture conditions for cellular homeostasis and forming a protective cushion that protects cell integrity and functionality during long-distance shipments and up to seven days.
What do the current regulations establish on the basis of BCRP and MDR1 transporter tests?
According to regulatory agencies, the identification of P-gp and BCRP substrates and inhibitors during CNS drug development is a regulatory requirement to circumvent potential clinical drug-drug interactions and nonlinear pharmacokinetics.
Is it possible to add a testing service to the order?
As a general rule, we act as a supplier and do not provide testing services. Nonetheless, feel free to contact us if you wish to test our plates externally. We can direct you to our partners who can assist you in conducting the assay effectively. Additionally, in certain situations, we are open to collaborating to try out new applications of interest to both of us.
Related news
A pioneering investigation unveils novel allosteric inhibitors of SHP2 in cancer therapy
In a recent publication in the Journal of Medicinal Chemistry, a collaborative research led by the scientist Dr. H. Day, in conjunction with a consortium of researchers, explored the development of allosteric inhibitors targeting SH2 domain-containing protein tyrosine...
Promising results for a new treatment against drug-resistant tuberculosis
The article highlights the discovery of a series of azetidine derivatives, known as BGAz compounds, that exhibit potent bactericidal activity against both drug-sensitive Mycobacterium tuberculosis and multidrug-resistant TB strains.
New Insights into drug formulation with ready-to-use Caco-2 cells
In a recent study published in the Future Pharmacology Journal, researchers from prestigious French companies examined the impact of four polyols on the permeability of seven active pharmaceutical ingredients (APIs).