ReadyCell News
Stay up-to-date on the latest ReadyCell news, projects and developments.
A ReadyCell & SOLVO Biotechnology meeting is taking place in Barcelona
We are happy to have with us Csenge Barabás, Nóra Kovács, Joseph Zolnerciksand and Dávid Bajkó to discuss a range of topics and to visit our facilities.
ReadyCell at the IX BiotechHealth Annual Symposium
ReadyCell will support academic research through its sponsorship at the IX BiotechHealth Symposium, which will be held on 28 June in Porto.
Cell-based in vitro models to characterize Food-Drug Interactions
It is well known that food intake changes luminal conditions (e.g. pH, motility, microbiota,…) in the stomach and the small intestine, modifying drugs bioavailability. Food-drug interactions are one of the major challenges for oral-administered drugs, even more so if considering the growing use of food supplements and functional foods.
1000 CacoReady batches
Congratulations to the manufacturing department for the comitmment and expertize in cell culture.
ReadyCell is expanding boundaries
The cell-based in vitro kits are shipped to South America, ensuring the quality standards. The patented biotechnology Shipping Medium® and the specialized logistic procedure at room temperature, achieve the proper delivery despite the long distances. ReadyCell is constantly growing and reaching out to more and more countries around the world.
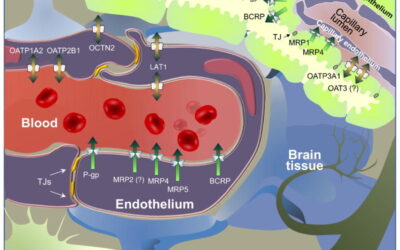
Assessment of Drug Permeation across the Blood-Brain-Barrier (BBB) in preclinical research
The endothelial cells that conform the BBB limit the passage of blood circulating endogenous substrates and larger molecules to the brain. This intrinsic protective role is also a major hurdle for reasearchers developing drugs for Central Nervous System (CNS) disorders.
CacoReady in the literature
Evaluate intestinal inflammation with Caco2 cell-based in vitro kits to reproduce phisiological conditions.
Review of available cell-based in vitro models to evaluate intestinal permeability
Cell-based in vitro models are suitable to evaluate human phisiology optimizing research. This review introduce reference models to evaluate gastrointestinal metabolism.
ReadyCell visits FarmaForum
Our collaborators Carla Sapienza and Marta Ollé are attending Farmaforum Madrid to discuss last updates in pharmaceutical and cosmetics research.
Drug Drug interaction studies in preclinical stage, reduces unexpected adverse events rate in clinical trails
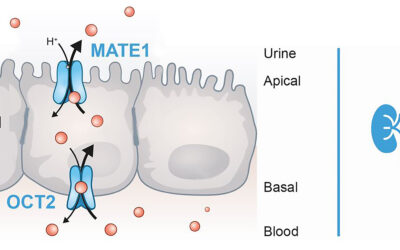
Specialized manufacturer of in vitro kits to evaluate uptake transporters, OCT2 and MATE1.
The evaluation of drug interactions in preclinical stages optimizes novel drug development
Specialized manufacturer of in vitro kits indicated to evaluate pharmacological relevant efflux and uptake receptors for TDI assays.
Role of pharmacological efflux receptors in cancer treatment resistance
Transmembrane efflux receptors, Pgp, MRP1 and BCRP has shown a relevant effect in the chemoterapy resistance.
Recommended cell-based in vitro testing to assess BCRP protein-mediated transport
PreadyPort BCRP is a in vitro system based on transfected MDCKII cells overexpressing the BCRP transporter.
3rd SCI-RSC Symposium on Transporters in Drug Discovery and Development
ReadyCell scientists shared their expertise ane experience on the impact of pharmacologically relevant drug-drug interactions.