
Jul 12, 2022
Last month, the ICH launched a draft on Drug Interaction Studies regulatory. The guideline includes the regulations of the European, American and Japanese medical agencies and covers both in vitro and clinical studies.
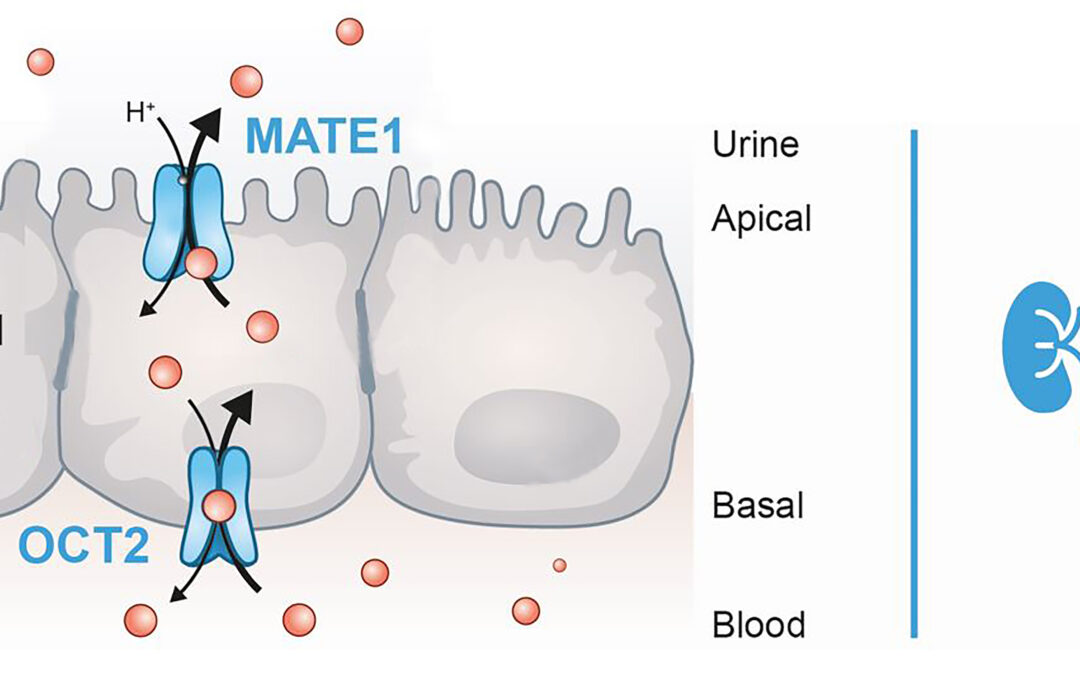
Oct 26, 2021
Specialized manufacturer of in vitro kits to evaluate uptake transporters, OCT2 and MATE1.
Sep 30, 2021
Specialized manufacturer of in vitro kits indicated to evaluate pharmacological relevant efflux and uptake receptors for TDI assays.

Jun 29, 2020
In vitro systems based on HEK293 cells are a recommended technique to assess MATE1 transporter interactions during preclinical stages.

Feb 28, 2020
Among other things, the agency explains that DDIs are a critical factor in a drug’s overall benefit-risk profile.






