ReadyCell News
Stay up-to-date on the latest ReadyCell news, projects and developments.
ReadyCell presented its latest software implementation at the Exact Event in Barcelona
This software was specially designed to manage our customers’ orders, as well as to automate the management of documentation and quality controls, facilitating orders worldwide and speeding up the generation of the necessary documents for customs.
A pioneering investigation unveils novel allosteric inhibitors of SHP2 in cancer therapy
In a recent publication in the Journal of Medicinal Chemistry, a collaborative research led by the...
ReadyCell at the Bio-Europe Spring 2024
ReadyCell is attending BIO-Europe Spring in Barcelona. The event is expected to bring together executives from biotech and pharma companies.
Happy International Women’s Day!
With this infographic, we proudly show the key facts we have achieved in recent times in promoting gender equality and women’s rights in the company.
Promising results for a new treatment against drug-resistant tuberculosis
The article highlights the discovery of a series of azetidine derivatives, known as BGAz compounds, that exhibit potent bactericidal activity against both drug-sensitive Mycobacterium tuberculosis and multidrug-resistant TB strains.
Get to know our talented women from Readycell
In honor of the International Day of Women and Girls in Science, we present a series of videos featuring insights from our team members that explore the diverse experiences and perspectives of the talented women at ReadyCell.
New Insights into drug formulation with ready-to-use Caco-2 cells
In a recent study published in the Future Pharmacology Journal, researchers from prestigious French companies examined the impact of four polyols on the permeability of seven active pharmaceutical ingredients (APIs).
Happy Holidays and a Prosperous New Year!
To convey our spirit, we share a Christmas card, proudly supported by ASHOFI, dedicated to aiding individuals affected by fibromyalgia.
3DLIVER: Celebrating one year of progress in the project for advanced drug toxicity testing and discovery
The main aim of the 3DLIVER project is the generation of innovative human liver-derived 3D in vitro models as innovative ready-to-use kits for drug testing models, using human liver cells and extracellular matrix (ECM), to generate more reliable in vitro drug testing devices.
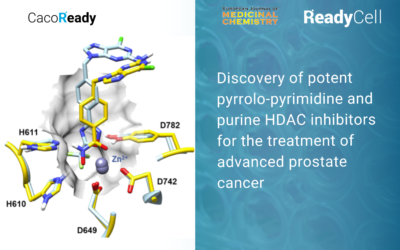
A new study that uses CacoReady reveals a potent inhibitor for prostate cancer
Evaluating in vitro permeability with CacoReady plates was central to the study design. By quantifying the permeability of the synthesized compounds within Caco-2 cells, researchers could identify and select which compounds were best in terms of cellular adsorption.
ReadyCell at the XV Spanish Drug Discovery Network Meeting in PRBB
ReadyCell is pleased to participate in the XV meeting of the Spanish Drug Discovery Network (SDDN) at the Parc de Recerca Biomèdica de Barcelona (PRBB) on November 20th and 21st, 2023.
Japanese distributor OYC meets with ReadyCell in Barcelona
The meeting focused on exploring the current market dynamics and emerging trends in the field of cell culture technology and drug discovery. Both companies, ReadyCell and OYC, are committed to providing their customers with top-tier products and services.
ReadyCell attends the CPHI Barcelona 2023
From October 24th to 26th, members of ReadyCell’s Business Development (BD), Research and Development (R&D), and Marketing departments attended the CPHI Barcelona 2023 Congress.
Seminar: What is Innovation?
During his talk, he explored the essence of innovation, its various forms, and its crucial role in driving growth and success.